Research
The central theme in our lab is to understand how mammalian cells survive and die in normal and diseased situations. We have focused on neurons but are also examining these pathways in stem cells, and in mouse models of neurodevelopmental disorders and neurodegenerative disease.
Our current projects are focused on the following four themes:
Apoptosis in neurons: How do neurons ensure their long-term survival?
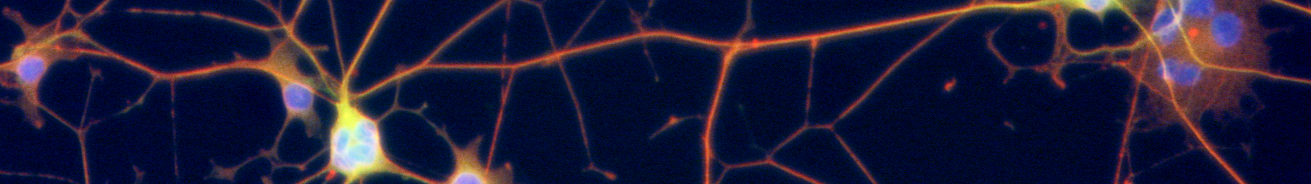
We and others have found that the apoptotic pathway is highly restricted in neurons. While the ability to undergo apoptosis in mitotic cells is important to prevent cancers, the robust restriction of apoptosis in postmitotic neurons is important to enable their long-term survival. For example, we have found that neurons engage multiple brakes in the apoptotic pathway to prevent caspase activation even after the point of mitochondrial permeabilization and cytochrome c release.
In current projects, we are investigating important survival mechanisms engaged by neurons to ensure their long-term survival, especially as the young neurons become mature and fully integrated in the neural network.
Axon pruning: How do neurons activate a degenerative pathway with precision in only the targeted axons?
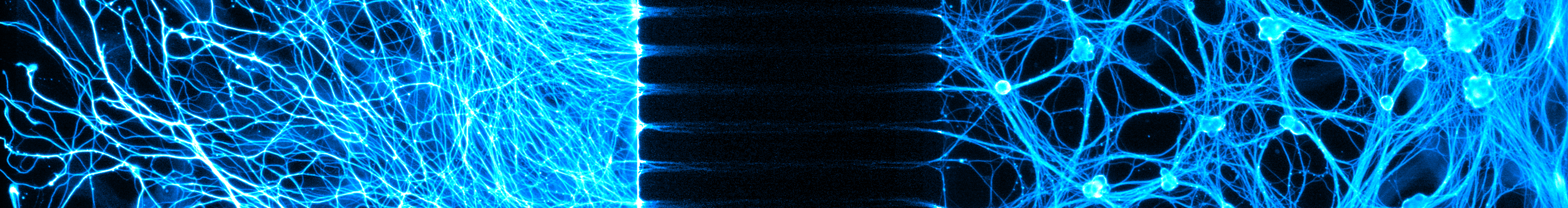
Neurons are able to selectively target specific axon segments for degradation via the physiological process of axon pruning. Axon pruning is important for the brain to maintain plasticity throughout life. One of the unexpected discoveries about axon pruning is that this pathway engages some of the same proteins that are activated during apoptosis (e.g. Bax, Caspases). Exactly how neurons are able to activate the apoptotic proteins in the context of axon pruning, where neurons survive, is unclear.
In current projects, we are using microfluidic chamber devices to compare the pathways of apoptosis and axon pruning and have found striking differences in how Bax and caspases are utilized in apoptosis versus axon pruning. We are probing the mechanisms by which neurons are able to activate caspases safely and with precision during axon pruning.
Stem cell differentiation into neurons: How do apoptotic thresholds change with neuronal differentiation?
We have found that while primary mitotic cells are inherently sensitive to apoptotic insults, postmitotic cells with limited regenerative potential and long lifespan, such as neurons, cardiomyocytes, and myotubes, have acquired stringent mechanisms for restricting apoptosis. While mitotic cells are at continual risk of becoming cancerous and need to maintain their ability to die rapidly, our ability to maintain the long-term survival of postmitotic cells in the brain, heart and skeletal muscle is critical for normal physiological functions.
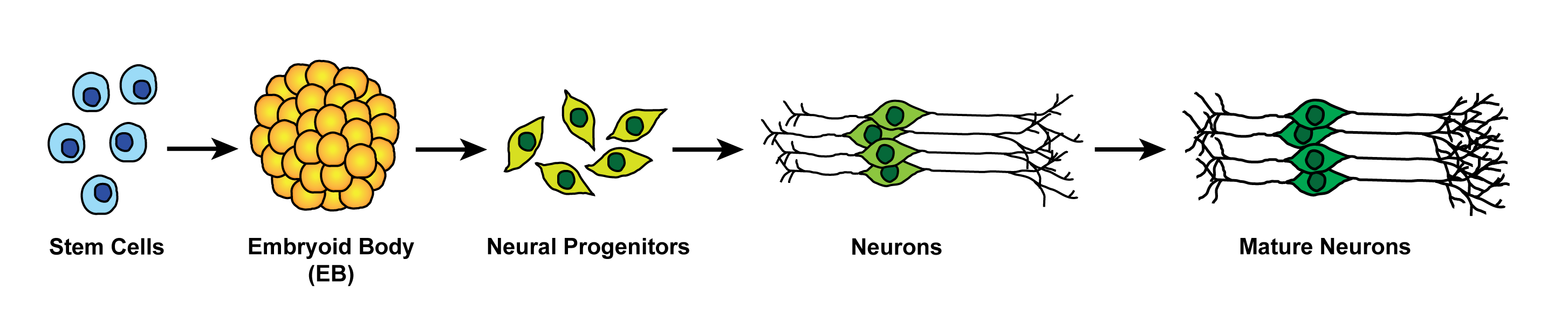
To examine how the survival and apoptosis thresholds dynamically change as mitotic cells become post-mitotically differentiated, we are using human Embryonic Stem (hES) cells induced to undergo neuronal differentiation. While the mitotic, undifferentiated hES cells are highly primed for rapid apoptosis, these cells become markedly resistant with neuronal differentiation. We are conducting various discovery screens to identify the events that are coupled with neuronal differentiation that dynamically change the thresholds of apoptosis. An exciting, and completely unexpected, outcome of this project is that we are discovering novel functions of known apoptotic proteins during neuronal differentiation.
How does the mature brain maintain homeostasis? A novel approach to develop therapeutics for brain disease
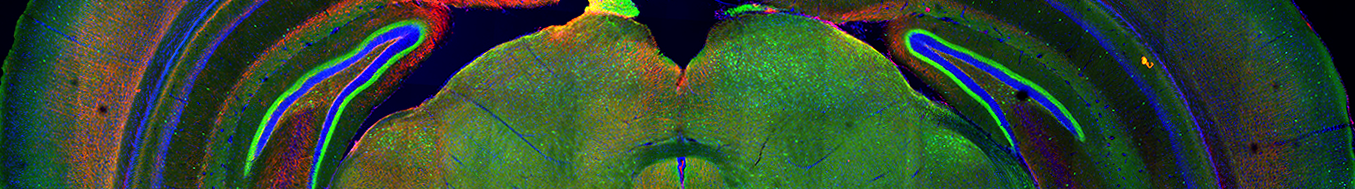
While research on how the brain develops and how it degenerates in disease have received considerable attention, the mechanisms that enable the mature brain to maintain homeostasis and survive long-term remain largely unexplored. This overall strategy of focusing first on identifying how the healthy brain maintains functional resilience and utilizing those biological molecules to restore function in the diseased brain could be a transformative approach for tackling neurodegeneration.
We have conducted screens to identify molecules that are engaged by the mature brain to enable its long-term homeostasis. In our current projects, we have focused on one such molecule we identified- the miR-29 microRNA. We have developed knock-out and overexpression mouse models of miR-29 and are finding that miR-29 has multiple essential function in the mature brain. Importantly, we are also evaluating the therapeutic capability of miR-29 in restoring brain homeostasis in the context of Alzheimer’s disease.